Working Papers
Publications in Economics
Publications in Health Policy
Maini, Luca, Rachel E. Sachs, Michael Anne Kyle, and Pragya Kakani. “Medicaid’s GENEROUS Most-Favored-Nation Pricing Model — Promise and Limitations.” New England Journal of Medicine 394, no.18 (2026): 1772-1773
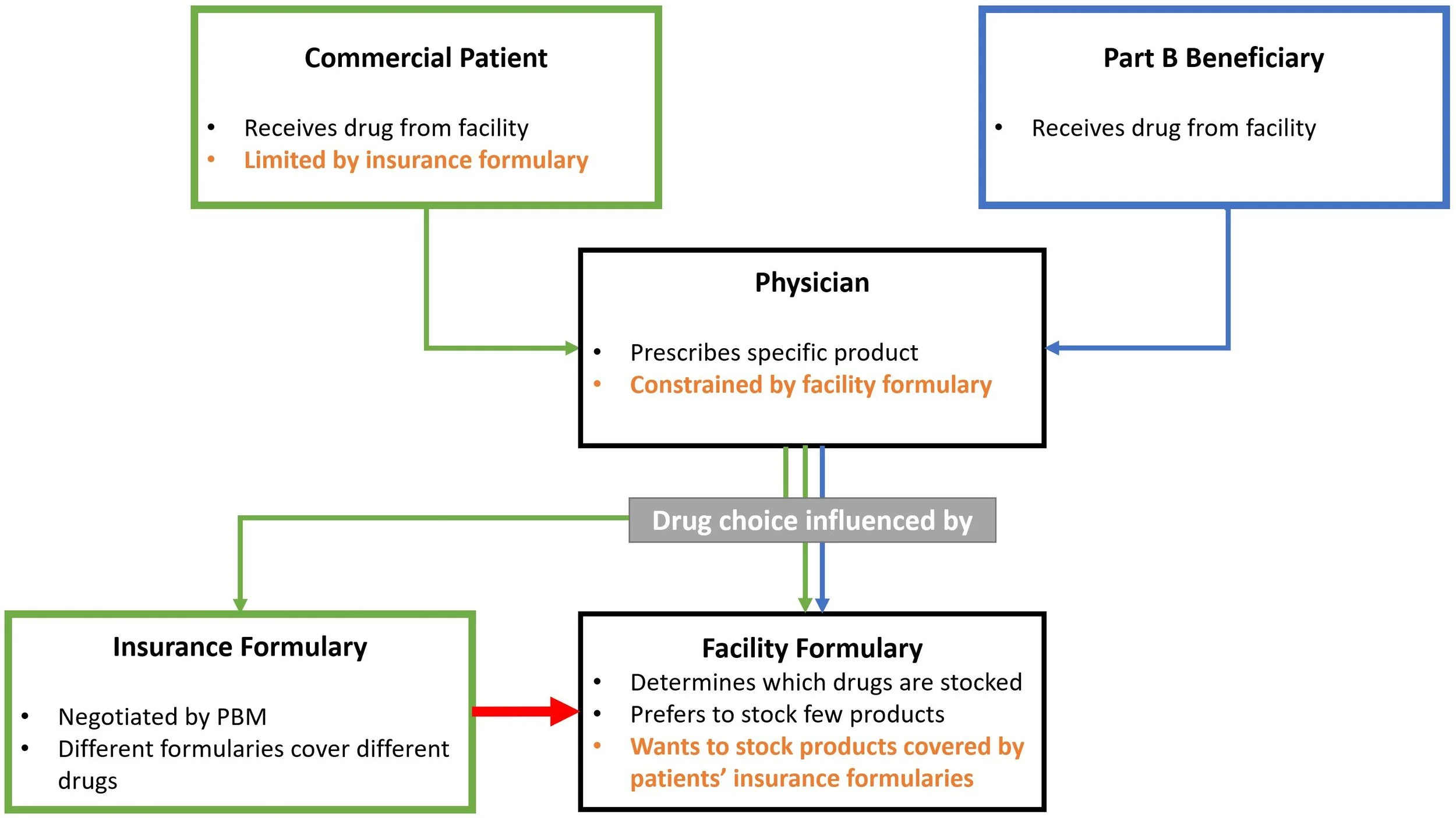
Kyle, Michael Anne, Pragya Kakani, Samantha Burn, and Luca Maini. “Trends in Prescription Drug Coverage Restrictions in Medicare, Medicaid, and Commercial Insurance Plans, 2011-2019” Health Affairs Scholar (2026): qxag059.
Axelsen, Kirsten, Delphine Courmier, Ge Bai, and Luca Maini. “Savings from biosimilars and Medicare formulary access.” Health Affairs Scholar 3, no. 11 (2025): qxaf214.
Kakani, Pragya, Bradley Katcher, and Luca Maini “Improved Insurance Coverage Increased Biosimilar Semglee’s Market Share After The FDA’s Interchangeability Designation.” Health Affairs 44, no. 11 (2025): 1417-1425
Benjamin N. Rome, Katherine Garrett, and Luca Maini. “Medicare Part D Savings Under the Manufacturer Discount Program vs Coverage Gap Discounts.” JAMA Network Open, 8, no. 9 (2025): e2530778.
Hwang, Thomas J., and Luca Maini. “Challenges in Delivering Most-Favored-Nation Pricing for Prescription Drugs in the US.” JAMA 334, no. 9 (2025): 763–764.
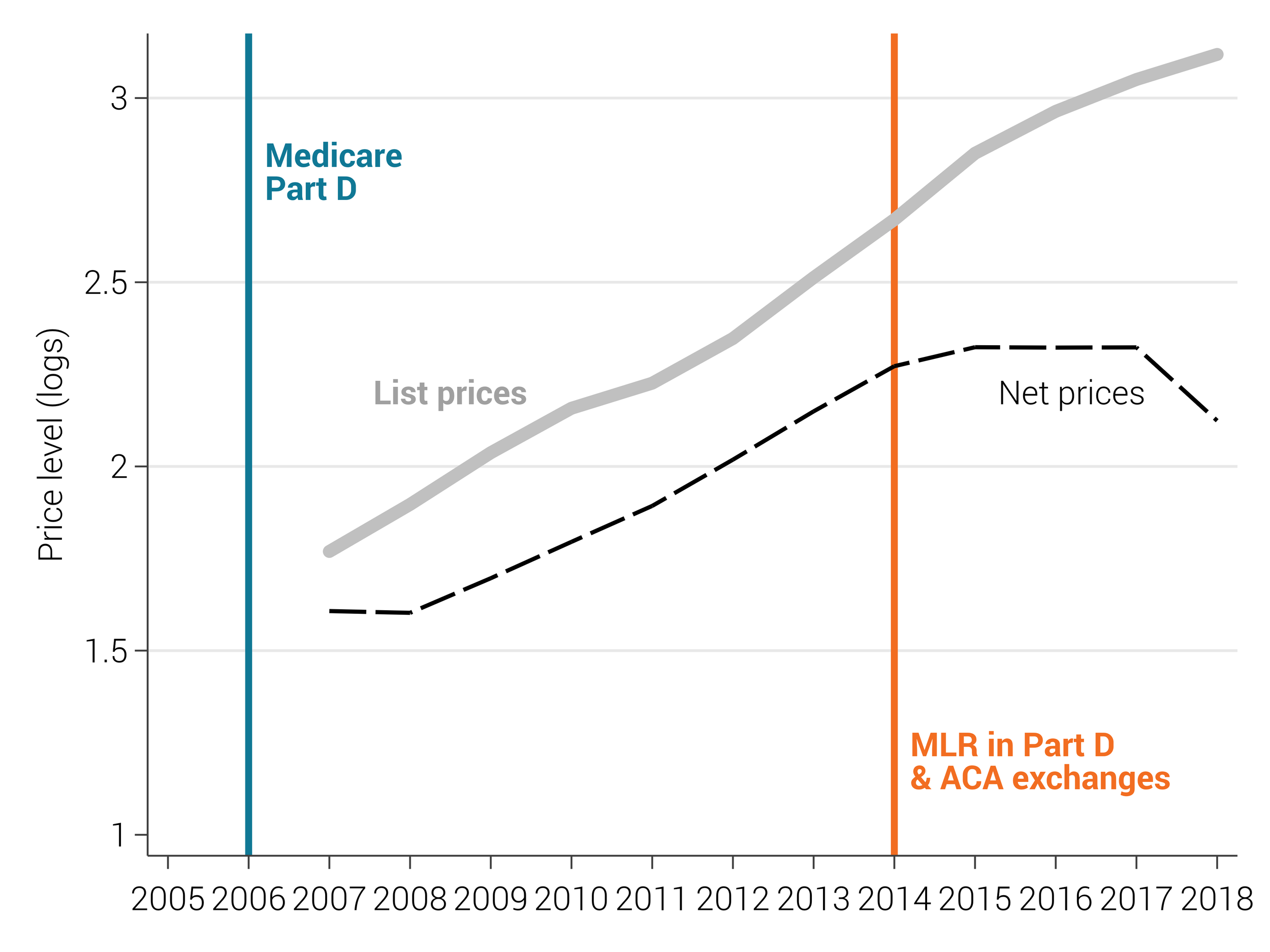
Kakani, Pragya, Michael Anne Kyle, Amitabh Chandra, and Luca Maini. “Medicare Part D Protected-Class Policy Is Associated With Lower Drug Rebates.” Health Affairs, 43, no. 10 (2024): 1420-1427
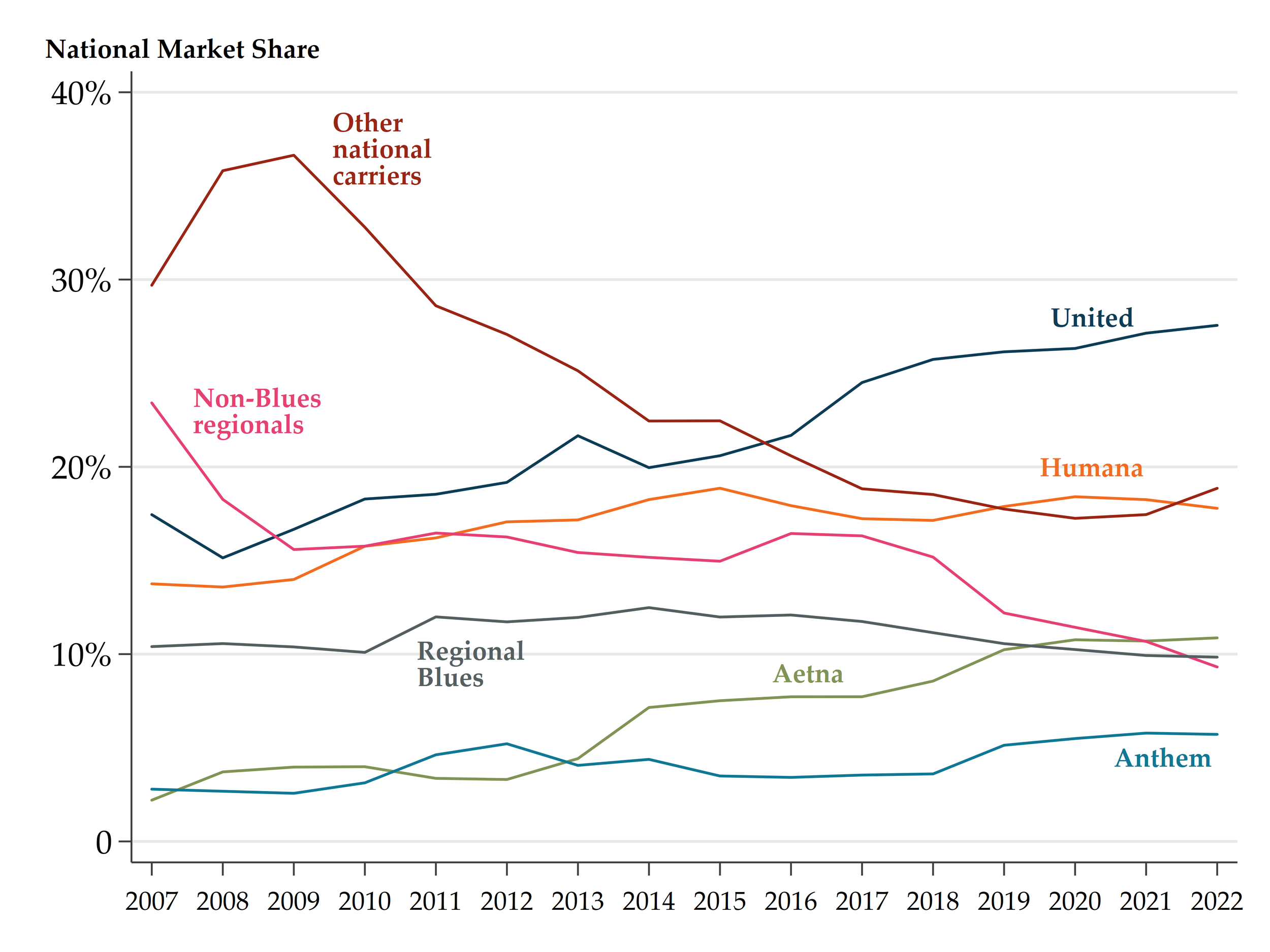
Bertuzzi, Luca, and Luca Maini. “Benefit Design And Biosimilar Coverage In Medicare Part D: Evidence And Implications From Recent Reforms.” Health Affairs, 43, no. 5 (2024): 717-724
Media coverage: PharmaVoice
Feng, Kimberly, Massimiliano Russo, Luca Maini, Aaron S. Kesselheim, and Benjamin N. Rome. “Patient Out-of-Pocket Costs for Biologic Drugs After Biosimilar Competition.” In JAMA Health Forum, vol. 5, no. 3, pp. e235429-e235429. American Medical Association, 2024.
Maini, Luca, Jacob P. Klimek, David C. Johnson, George M. Holmes, and Darren A. DeWalt. “Impact of Including Drug Spending in Oncology Alternative Payment Models” The American Journal of Managed Care 29, no.11 (2023): 579–584.
Hwang, Thomas J., Josh Feng, Luca Maini, and Aaron S. Kesselheim. “Medicaid Expenditures and Estimated Rebates on Line Extension Drugs, 2010–2018” Journal of General Internal Medicine 37, no. 14 (2022): 3769-3771.
Other Writing
Maini, Luca. “Pharmaceuticals,” in the Elgar Encyclopedia on the Economics of Competition, Regulation and Antitrust, Michael D. Noel (Editor). Cheltenham, UK: Edward Elgar Publishing (2024).
Maini, Luca. “La regolamentazione dei prezzi dei farmaci: lezioni da recenti avanzamenti empirici, “ in Il Prezzo dei Farmaci, Fabio Pammolli (Editore). Bologna, IT. Società editrice il Mulino (2024) [english version]
Feng, Josh, and Luca Maini. “Where is the Good Stuff? Drug Quality Measures and Economic Research.” NBER-IFS White Paper, 2016.